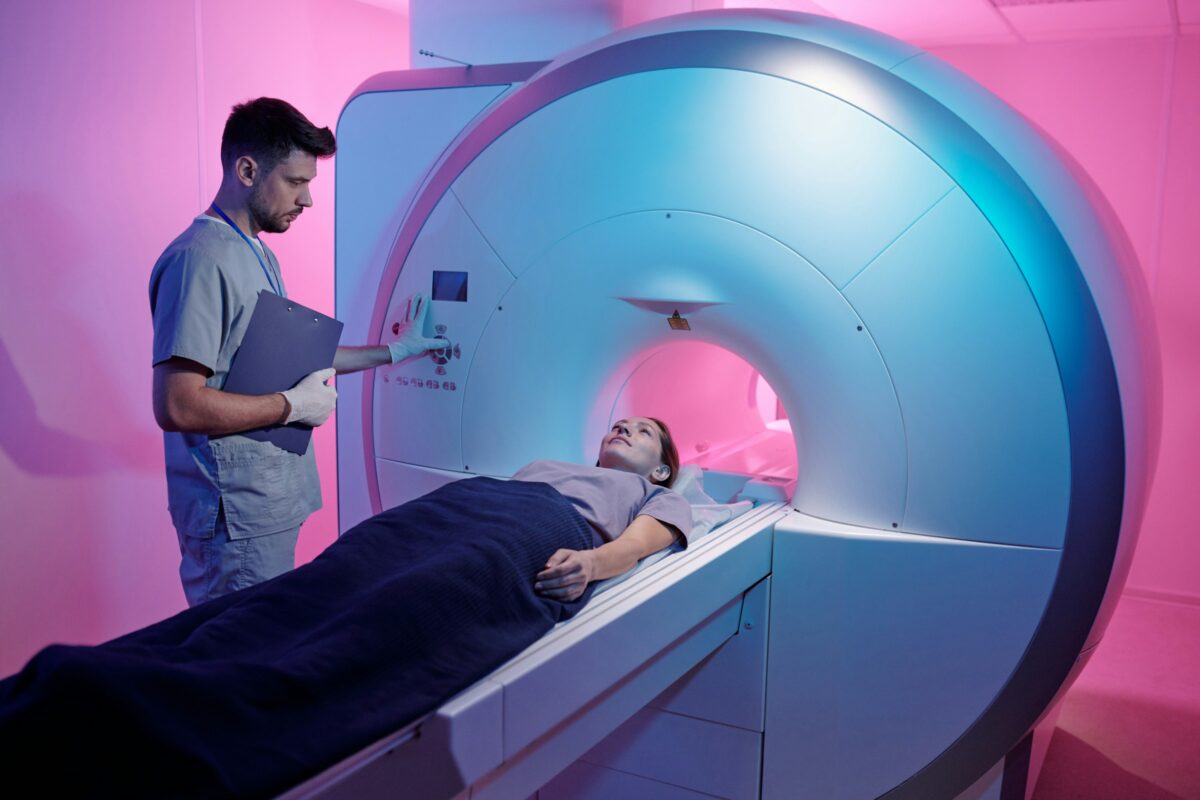
K2 is recruiting for a new Phase 3 trial for Alzheon’s new drug, which aims to be the first oral disease-modifying therapy for Alzheimer’s.
Recently, K2 started screening possible trial participants for a Phase 3 clinical trial of a new Alzheimer’s drug. This drug, called valiltramiprosate, is designed to prevent protective forms of beta amyloid from sticking to each other and becoming toxic forms. These plaques gum up the works in the brain and lead to the death of brain cells. K2’s ongoing APOLLOE4 trial will assess whether valiltramiprosate can actually slow down cognitive decline in people who carry genetic risk factors for Alzheimer’s.
While scientists don’t know yet what causes Alzheimer’s, beta-amyloid protein plaques remain one of the prime suspects. While many approaches have focused on clearing these amyloid plaques, this strategy has not yet succeeded in bringing disease-modifying treatments to market. So far, most existing treatments for Alzheimer’s are only designed to help people manage symptoms, like memory loss.

WHAT IS ALZHEON’S NEW DRUG? ![]()
Valiltramiprosatee, an experimental Alzheimer’s drug by Alzheon, is taking a different approach. It protects a healthy form of amyloid-beta from turning into a more toxic form. The drugmakers theorize that if they can prevent these proteins from sticking together, they could prevent the damage that occurs in brains with Alzheimer’s.
Another big difference when it comes to valiltramiprosate is that — unlike the sole FDA-approved disease-modifying drug available (Aduhelm) and those close behind it in the pipeline to hopeful FDA approval (like lecanemab) — valiltramiprosate is taken orally. This means that if valiltramiprosate succeeds in clinical trials and lands FDA approval, it will be easier to administer. Aduhelm and the several experimental antibody-based treatments underway all require intravenous infusions, which are harder to administer, more expensive, and less accessible.
WHO IS ELIGIBLE FOR THIS ALZHIEMER’S DRUG?![]()
K2 is currently recruiting for the Phase 3 trial, specifically for participants aged 50 to 80 years with progressive memory loss over the past 12 months. They will be screened to see if they have two copies of a gene which confers the highest risk for Alzheimer’s: APOE4.
So, why the focus on APOE4? Most people who carry two copies of this gene can increase the risk of developing Alzheimer’s by up to 12 times. This gene is linked to pathogenic protein deposits that disrupt brain function in Alzheimer’s as well as cardiovascular health. So, if a treatment that targets Alzheimer’s is going to work for anyone, the trial designers figure, it is most likely to work on people with the highest risk of developing Alzheimer’s.

HOW WILL THE TRIAL WORK?
In studies for infused anti-amyloid drugs like Aduhelm and lecanemab, more than 20 percent of patients experienced a form of brain swelling as a side effect. So far, valiltramiprosatee does not appear to cause these side effects which require extra brain scans and monitoring.
Since the screening procedures involve testing for genetic risk factors, the K2 team estimates it may take up to 13 weeks to learn whether you are eligible for the study. The screening process involves coming to the site, signing a consent form, completing a short memory assessment, and getting blood drawn to see if you qualify.
Once enrolled, you will be given the treatment or placebo to be taken twice daily, for approximately a year and a half. Subsequent study visits will involve other procedures to check heart health, vitals, neurological and physical functions, and cognition, to establish if the drug results in substantial cognitive improvement. All together, the study only requires nine visits to the K2 center for testing.
The principal investigator for the trial is Dr. Brandon Lenox. Call K2 if you’re interested in participating: 407-500-K2K2 (5252).